
Claims and Description
Most patent laws establish that the right conferred by a patent is determined by its claims (see, for ex., Section 11 of the Argentine Patent Law, Articles 69 and 84 of the European Patent Convention (EPC), or paragraph 112(b) of Title 35 of the United States Code). Thus, the claims define the subject matter for which protection is sought (namely, the invention).
For this reason, the proper drafting of patent claims is of vital importance to an intellectual property strategy: the scope of the claim will be determined by the terminology used and will define which embodiments are covered.
For example, if a claim uses the term “vinyl polymer,” its scope will be broader than if it used the term “polystyrene” (a specific type of vinyl polymer). While this results in broader protection, it also increases the number of prior art documents that may affect patentability, as the scope of protection is inversely related to patentability.
Now, if the claims define the right and therefore constitute the “heart” of a patent, what is the role of the description? In short, the description must support the claims, and describe the invention defined by them in a sufficiently clear and complete manner.
On the other hand, patent laws establish that the description, which includes a written description and, optionally, drawings, is used to interpret the claims. In general, it is the patent offices or courts that decide how this interpretation is to be carried out. Moreover, these interpretations may differ from one another, as is the case in the United States (the “broadest reasonable interpretation” standard before the USPTO vs. the “ordinary and customary meaning as understood by a person of ordinary skill in the art in light of the description and prosecution history” standard applied by the courts).
It is also common for the same right holders to advocate for different interpretations of the same claims, depending on whether the goal is to assess the scope of the right in the context of infringement, or to analyze whether the invention meets the patentability requirements. In the former case, the right holder will tend to argue that the claim has a broad scope, allowing them to block a larger number of competitors, while in the latter, they will argue for a narrower interpretation to strengthen the patentability position.
In this context, a key question arises: what happens when the terms of a claim may be interpreted in a certain way “on their own,” but are defined or given a proposed interpretation within the description?
This question was recently addressed by the Enlarged Board of Appeal of the European Patent Office (EPO) in its decision G 1/24. This decision originated from case T 0439/22, in which the term “gathered sheet” had to be interpreted in the claims of patent EP 2 307 6804.
The usual interpretation would be “a sheet folded along lines to occupy a three-dimensional space.” However, the application indicated that the term should be interpreted as “corrugated, folded, or compressed or otherwise constrained substantially transversely to the cylindrical axis of the rod.” Using this latter interpretation, patentability—particularly novelty—was affected by prior art. For this reason, the applicant sought to distance itself from the interpretation proposed in the description, arguing that claim terms should be interpreted according to their usual meaning and that the description should be consulted only in case of doubt.
Three questions were posed to the Enlarged Board in case T 0439/22, including:
Can the description and drawings be consulted when interpreting the claims to assess patentability and, if so, can this be done as a general rule or only if the person skilled in the art finds the claim unclear or ambiguous when read in isolation?
In response, the Enlarged Board considered that there is no single legal basis for claim interpretation in the context of assessing patentability, as Article 69 of the EPC relates to the scope of protection for infringement purposes, while Article 84 sets formal requirements for the content of a patent application and does not refer to claim interpretation.
Nevertheless, based on the case law of the Technical Boards of Appeal, the Enlarged Board concluded:
“The claims are the starting point and the basis for assessing the patentability of an invention. The description and drawings shall always be consulted to interpret the claims when assessing the patentability of an invention, and not only if the person skilled in the art finds a claim to be unclear or ambiguous when read in isolation.”
This decision reaffirms the importance of the description in interpreting the claims in all circumstances. It also sets out guidelines for the alignment between the description and the claims —something that some EPO examiners were already strictly enforcing when requesting amendments to the description in line with the allowable claims following examination.
Decision G 1/24 may influence how courts, both in Europe and other jurisdictions, interpret the scope of protection of a granted patent, and reinforces the importance of precise drafting to protect the interests of right holders.

Taylor Swift, Her Masters, and Intellectual Property as a Strategic Asset
Recently, Taylor Swift made global headlines when she announced that, after years of litigation, she had repurchased the rights to the original recordings of her first six albums — known in the music industry as masters. The origins of the conflict date back to 2004, when Swift signed her first record deal with Big Machine Records at the age of 14. As is often the case in early contracts with emerging artists, she gave up ownership of those recordings in exchange for the support she needed to launch her career.
In 2018, when her contract ended, Swift attempted to regain control over those works. But Big Machine Records was acquired by entrepreneur Scooter Braun, who bought the entire catalog — including the masters — without giving Swift the opportunity to be involved in the deal. Then, in 2020, Braun sold the recordings to an investment fund, once again without offering the artist the chance to match the offer.
In response, the singer-songwriter decided to re-record her albums under the label “Taylor’s Version,” thereby weakening the commercial value of the original versions and generating the resources to buy back the rights to her masters. This was possible because her original contract included a clause allowing her to re-record her albums after a certain period following its expiration.
Beyond the media buzz, this case highlights how intellectual property assets — intangible yet strategic — are essential tools for protecting creativity, innovation, and their capitalization potential. Effective management of these assets requires an interdisciplinary approach that integrates technical, legal, commercial, and regulatory aspects. Simply registering an asset is not enough: it is crucial to accompany its life cycle, anticipate technological, regulatory, and business shifts, and design strategies to maximize its long-term value. In this regard, specialized and continuous advice is key for those aiming to transform creativity and innovation into a sustainable business while maintaining control over their future.
In the wake of Taylor Swift’s case, more artists may opt to license their masters rather than sell them outright. For their part, record labels are likely to become more reluctant to allow re-recordings of acquired masters, in order to prevent other artists from replicating the strategy employed by the American singer-songwriter.
Taylor Swift’s story shows that managing intangible assets is not a one-time action, but a long-term strategy. From registering her name as a trademark in 2008 to building a portfolio that includes song titles, iconic phrases, and elements from her narrative universe, she has successfully developed a personal brand with enormous commercial power.
In a context where innovation is accelerating and business models are becoming increasingly complex, intellectual property serves as a framework that structures, protects, and enables development. It is not merely a legal formality — it involves building assets that allow knowledge to be monetized, scaled sustainably, and strategically controlled as the business evolves.
In an increasingly globalized, digital, and dynamic world, holding intellectual property assets opens doors to new business opportunities. Proper management of these assets facilitates the creation of strategic alliances, access to international markets, and negotiation of licenses that boost revenue generation. Thus, IP becomes a competitive tool that can make the difference between the success or stagnation of an innovative project.
Whether we are talking about songs, software, chemical formulas, industrial developments, or trademarks, the message is the same: understanding, planning, and strategically protecting intellectual property assets is a necessary condition for leading the creative and commercial destiny of any project.

The Unified Patent Court (UPC) in Europe issues judgments related to biotechnological inventions
With the relatively recent establishment of the Unified Patent Court (UPC) in Europe, which has jurisdiction over the territories of the European countries that have signed the Agreement on a Unified Patent Court (UPCA), there is great anticipation among professionals in the field of industrial property regarding the judgments issued by this Court, that will lay the foundation for the precedent to be followed in patent litigation across the European continent in the years to come.
In this regard, the Court of First Instance of the UPC issued two judgments in July of this year related to patents in the field of biotechnology, a field of great relevance today and one that represents a particularly thriving and innovative industry in our country. Although the judgments in question are not final, as they are first-instance decisions and therefore subject to appeal, it is worthwhile to analyze the Court’s considerations when reaching its decisions.
This judgment addresses a preliminary injunction requested by the plaintiff Amycel LLC (hereinafter, “Amycel”), against the alleged infringer of its European patent EP 1 993 350 B2, which claims a hybrid strain of the fungus Agaricus bisporus. This strain is defined in the claim by its deposit number in the American Type Culture Collection (ATCC), a recognized biological material depository institution in accordance with the Budapest Treaty.
The alleged infringer, a Polish farmer engaged in mushroom cultivation and commercialization, marketed a mushroom of the Agaricus bisporus species in the Netherlands—a country under the UPC’s jurisdiction—under the brand name “Cayene,” which Amycel claims infringes its EP 1 993 350 B2 patent. As a result of this commercialization, Amycel sought a preliminary injunction against the alleged infringer before the UPC, requesting the Court to order the alleged infringer to cease the unauthorized use, withdraw the infringing products, and pay damages.
The judgment text emphasizes that both parties agreed that the condition for infringement to exist was that the allegedly infringing product must be genetically identical to the strain claimed in the patent. The difference in the positions adopted by the parties lies in the interpretation of what it means for both organisms to be genetically identical: the defendant argued that for genetic identity to exist between the Cayene product and the BR06 strain, both products must exhibit 100% genetic similarity. Amycel, however, contended that due to technical and biological factors, it is inevitable that even two identical samples will yield a genetic sequencing result of less than 100% identity, supporting its argument with experimental data.
Considering that the experimental results presented by both parties indicate that the observed genetic similarity between the disputed product and the claimed strain falls within the expected margin of error for identical samples, the Court held that it is more likely than not that the commercialization of Cayene infringes EP 1 993 350 B2. Consequently, it found no reason to dismiss the request for provisional reliefpreliminary injunction and ordered the defendant to cease commercialization and withdraw the infringing product from the market.
Based on the analysis of the “Amycel” judgment, it can be concluded that, in the eyes of the UPC, it is clear that for there to be an infringement of a patent protecting a specific strain of an organism deposited in an institution such as the ATCC, experimental results obtained by competent third parties, as well as the opinion of experts in the field interpreting those results, are of fundamental importance in determining the existence of genetic identity between the products in dispute.
- “Sanofi v. Amgen” judgment
This judgment addresses a request for the invalidation of patent EP 3 666 797 B1, held by Amgen, Inc. (hereinafter, “Amgen”), filed by a group of companies from the Sanofi group (hereinafter, collectively referred to as “Sanofi”) within the framework of an infringement lawsuit initiated by Amgen against Sanofi.
The patent claims a monoclonal antibody defined by functional features, which are essentially:
- that the antibody is for use in the treatment or prevention of hypercholesterolemia, or to treat atherosclerotic disease or reduce the risk of a cardiovascular event associated with elevated blood cholesterol levels;
- that the antibody binds to the catalytic domain of the PCSK9 protein with a specific amino acid sequence and prevents PCSK9 from binding to LDLR (a protein involved in cholesterol metabolism and blood cholesterol levels).
European practice allows for claiming antibodies defined by their “target,” that is, by the entity to which they bind. When defining the invention in this way, it is particularly relevant to demonstrate that the achieved technical effect results, on the one hand, results from the antibody’s binding to the specified target, regardless of the specific sequence of the antibody used, and on the other, that it is not obvious from the prior art.
In this case, the Court ruled that the claimed invention lacks an inventive step in view of a scientific article describing several experiments aimed at establishing the relationship between the PCSK9 protein and LDLR.
The article in question suggests that neutralizing PCSK9 activity, including developing antibodies to block its interaction with LDLR, could be useful in treating hypercholesterolemia. Therefore, the Court concluded that a person skilled in the art, considering the teachings of the cited article, would have been motivated to develop antibodies that block the interaction between PCSK9 and LDLR for the treatment of hypercholesterolemia. According to the Court, generating and selecting such antibodies would be a routine task for a skilled practitioner.
Notably, the Court did not consider the characteristic of the antibody binding to the catalytic domain of PCSK9 to contribute to an inventive step, as no causal technical link was demonstrated between this characteristic and the reduction of PCSK9-LDLR binding, making binding to the catalytic domain an arbitrary selection among several options.
This judgment is particularly interesting given that a parallel case in the U.S. also resulted in the patent being declared invalid, albeit on different grounds. The U.S. Supreme Court rendered the patent invalid not on the grounds of obviousness over prior art, but due to a lack of “enablement”. The Court found that the description provided in the patent did not enable a person skilled in the art to make and use the claimed invention across its full scope. Two key factors for the Court’s decision were: (i) the patent provides examples of the preparation and characterization of 26 antibodies, while its claims potentially cover millions of antibodies, and (ii) the instructions provided in the patent for obtaining antibodies beyond the 26 exemplified amount to, according to the Court, merely “research projects” that would force a person skilled in the art to undertake an undue level of experimentation to determine which potential embodiments of the invention achieve the intended purpose.
This judgment serves as a particularly relevant case study on how different jurisdictions interpret the requirements a patent must meet to be considered valid.
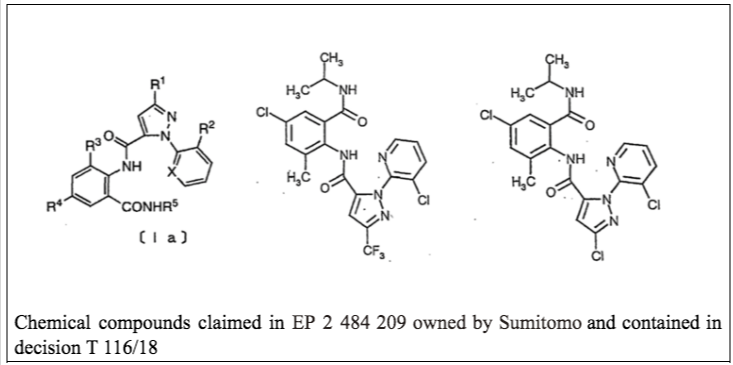
Plausibility before the European Patent Office
On March 23, 2023, decision G 2/21 was published regarding the “Plausibility” of an invention before the European Patent Office (EPO). According to the understanding of the EPO’s Enlarged Board of Appeal, the plausibility of an invention is related to the possibility of supporting a technical effect upon which the inventive step relies, through experimental data or evidence submitted after the filing of the patent application.
The filing of data or experimental results after the filing of a patent application is a common practice in the patent prosecution process. In general, these pieces of evidence of the inventive step are generally accepted when they support or clarify a result obtained through the invention. The acceptance of these pieces of evidence varies depending on the territory or technical area, but they are generally accepted when the technical effect being demonstrated is disclosed in the application as originally filed.
The Technical Boards of Appeal of the EPO, in several decisions, considered cases in which the technical effect was not explicitly disclosed but could be deduced or considered by a person having ordinary skill in the art based on the information of the patent application, regardless of their common general knowledge. In other words, they considered situations in which it was either credible or plausible, or it was not non-credible or implausible, that a certain technical effect could be obtained through invention disclosed in the application as originally filed.
In this context, decision T 116/18 referred three questions to the Enlarged Board. In summary, the questions aim to decide on the admissibility of evidence subsequently submitted when the technical effect is not explicitly disclosed in the application as filed, but when this effect would be considered plausible by a person having ordinary skill in the art based on the information of the patent application (ab initio plausibility), or when there is no reason for the skilled person based on the information of the patent application to consider this effect as implausible (ab initio implausibility).
To answer these questions, the Enlarged Board affirmed in G 2/21:
- Evidence submitted by a patent applicant or proprietor to prove a technical effect relied upon for acknowledgement of inventive step of the claimed subject-matter may not be disregarded solely on the ground that such evidence, on which the effect rests, had not been public before the filing date of the patent in suit and was filed after that date.
- A patent applicant or proprietor may rely upon a technical effect for inventive step if the skilled person, having the common general knowledge in mind, and based on the application as originally filed, would derive said effect as being encompassed by the technical teaching and embodied by the same originally disclosed invention.
With this decision, the EPO reaffirms its position regarding the evidence submitted after the date of filing of a patent application, and which intends to support a purported technical effect.
It is important to highlight that the Enlarged Board reminds that this approach is applicable for inventive step, but that according to the current practice of the EPO, it would not be applicable for the assessment of sufficiency of disclosure.
Furthermore, according to the Enlarged Board, plausibility is not a new requirement for patentability, as it is novelty, inventive step, industrial application, and sufficiency of disclosure, instead, it “rather describes a generic catchword seized in the jurisprudence” as a criterion to support a purported technical effect. The concept of plausibility would be already included and considered in all other requirements, for which is not necessary to amend the laws related to patentability.
There are some territories with very restrictive practices for accepting evidence submitted after the filing date of a patent application. For example, in Argentina, for the prosecution of patent applications in the pharmaceutical field, these subsequent submissions are generally not accepted. The effect that this new decision by the EPO will have on the practice of other territories remains to be assessed.

Patenting antibodies in Europe and Argentina
On March 1, 2021, the new Guidelines for Examination of the European Patent Office (EPO) came into force. These Guidelines set the rules by which Examiners at the EPO analyze the patent applications prosecuted therein to evaluate whether they are in conditions to be granted as European patents, thus providing more homogeneity and transparency to said prosecutions.
The new Guidelines include a section devoted to the patentability of antibodies, inexistent thus far, which essentially sets in stone the criteria by which the Examiners at the EPO have been conducting their examinations on applications related to antibodies lately. Said criteria are based in decisions issued in the last few years by the Technical Board of Appeals of the EPO on antibodies-related issues, used so far by the Examiners to carry out their analyses in lieu of specific indications in the Guidelines.
The new section of the Guidelines (section G-II-5.6), on one hand, establishes requisites for the proper definition of antibodies in claims, and on the other, provides considerations on how to demonstrate the inventive step involved in the development of novel antibodies.
Definition
Antibodies are proteins (i.e., polypeptides) and, as such, they structurally consist of a series of linear amino acids chains. They are part of the immune system of many living organisms, and every antibody is adapted to bind to a particular antigen (i.e., a substance which generates an immune response in the organism) by specific regions in its amino acids sequence named complementarity-determining regions (CDRs). Each antibody has 6 CDRs, which define to which antigen they will bind.
According to the new Guidelines, the EPO accepts defining a claimed antibody by either structural or functional features, as well as by its production process.
- Structural features The essential structural feature of antibodies is their amino acid sequence. Since the CDRs are the parts of an antibody which determine their ability to bind to a certain antigen, the EPO establishes that the structural definition of an antibody must include, at least, the amino acid sequences of the 6 CDRs thereof. The EPO will only accept a structural definition of an antibody including less than all 6 CDRs if it is unequivocally proven that not all CDRs are involve in the binding to the antigen. Another way to define an antibody structurally would be by means of the nucleotide sequence codifying it (i.e., the specific portion of a nucleic acid molecule, for instance DNA, which may be used by a cell to generate the corresponding antibody).
- Functional features The EPO contemplates the possibility of defining an antibody by several types of functional features, such as the antigen to which it binds, the affinity therefor it exhibits, the specific regions of the antigen to which it binds (named epitopes), etc., as well as combinations thereof. Even though this kind of definitions may be desirable because they are usually of a broader scope than structural definitions, it is important to note the new EPO Guidelines clearly establish that in these cases it is especially important for the Examiner to verify whether the specification of the patent application under analysis provides a sufficient description of the invention, so that it may by reproduced by a person of skill in the art (i.e., that the description of the invention allows for it to be reproduced without any ambiguity). On the other hand, the Guidelines clarify that, if there is any prior art disclosing an antibody directed to the same antigen as the one being claimed, which is also obtained by a process as the one used for the claimed antibody, it will be assumed that the prior art inherently disclosed the functional features by means of which the same is defined, which would deny its novelty.
- Production process The EPO allows defining an antibody by means of the process by which it is obtained. For instance, the antibody could be defined by the immunization protocol used to generate it. However, it should be noted that in these cases the antigen used for the generation of the antibody should be defined by its sequence without any ambiguity whatsoever.
Inventive step
Regarding inventive step, the new section of the EPO Guidelines establishes that a novel antibody which binds to a known antigen must exhibit an unexpected property over antibodies known in the prior art for the same antigen. Examples of such properties might be a greater affinity, a higher therapeutic efficacy, reduced toxicity or secondary effects, etc. A mere structural difference with the antibodies of the prior art would not be considered enough in this case to provide an inventive step, since such a difference would be considered an “alternative solution” to a technical problem previously solved, which the EPO considers obvious in view of the prior art as a general rule in this technical field.
Situation in Argentina
Even though the Argentinean PTO (INPI, National Institute of Industrial Property) included in its Patentability Guidelines a section directed to biotechnological inventions by means of Resolution INPI N° P-283 of September 25, 2015, they do not provide specific indications regarding inventions related to antibodies. However, there are some tendencies which Examiners at the INPI usually follow when analyzing this kind of applications.
Regarding the definition of claimed antibodies, the INPI only accepts a structural definition thereof. Unlike the EPO, neither a functional definition nor a definition by means of the production process for the claimed antibody are acceptable. This means that some inventions which would be potentially patentable in Europe would not be accepted in Argentina, or at least they would be accepted with a much more limited scope (for instance, having to limit a broad functional claim to a narrower one defined by the specific sequences of the claimed antibody).
For a proper structural definition of a claimed antibody, Examiners at the INPI usually require that all 6 CDRs of the antibody to be defined. On the other hand, defining an antibody solely by means of the nucleotide sequence which codifies it is generally not accepted, as it should be complemented specifying as well the codified amino acid sequence (as mentioned, al least the 6 CDRs).
Regarding the evaluation of inventive step, the INPI does not have a criterion as clear as the EPO in this aspect. However, as a general rule, Examiners at the INPI tend to expect a patent application to include comparative results to the closest prior art, showing any surprising effect. In the specific case of antibodies, this would imply a similar criterion to that of the EPO, which is why it would be appropriate to include results in the specification showing some unexpected advantageous property of the claimed antibody in comparison to those of the prior art directed to the same antigen.

Pat-INFORMED by WIPO
Pat-INFORMED, A PATENT INFORMATION TOOL FOR PHARMACEUTICALS
“Pat-INFORMED” (Patent Information Initiative for Medicines) by WIPO is a database containing information on granted patents of invention directed to small-molecule pharmaceutical drugs.
It is part of a program developed by several global research-based biopharmaceutical companies that voluntarily provide information to the database as holders of the corresponding patent rights. The program is supported by WIPO and the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA).
The database only contains information regarding granted patents of products that are marketed and/or approved in at least one country.
Patents are listed according to the drug international non-proprietary name (INN), allowing users a simple way of accessing data. This is a remarkable and advantageous feature for those not used to dealing with other patent databases.
Besides the information provided by the participating volunteers, WIPO provides supplementary information and links to other public databases, like WIPO’s Patentscope.
Pat-INFORMED database may be accessed through the following link: https://www.wipo.int/patinformed/

USPTO: New COVID-19 Provisional Application Pilot Program
USPTO Pilot Program enables deferred-fee payment related to filing of provisional patent applications that combat COVID-19
The United States Patent and Trademark Office (USPTO) launched a pilot program that allows deferring the payment of filing official fees for provisional patent applications that combat COVID-19, until the filing of a corresponding non-provisional application.
The applicants must agree that the technical subject matter disclosed in their provisional applications will be available to the public on USPTO’s website.
In this way, two objectives are achieved: by disseminating the technology, applicants contribute to the fight against COVID-19 while at the same time protecting their patent rights.
To qualify for the program, the subject matter disclosed in the provisional must concern a product or process related to COVID-19, and such product or process requires Food and Drug Administration (FDA) approval for COVID-19 use.
The program accepts applications as from September 17, 2020, for a duration of 12 months.
For more information, please click here.

WIPO PROOF: new “digital fingerprint”
Assists in proving the existence of innovations, trade secrets and confidential information
The World Intellectual Property Organization (WIPO) has launched a new “digital fingerprint” service that provides a certain time and date to digital files containing, for example, intellectual creations such as innovations, trade secrets, confidential information, research and data.
This “digital fingerprint” is conceived as a complement to existing IP protection systems (trademarks, patents, designs, etc.) and, particularly, as evidence to solve disputes related to the use of confidential information or certain innovations before they are formally registered as IP rights.
Although WIPO PROOF does not create IP rights, the possibility of demonstrating that certain content existed at a given moment may assist in protecting assets during each stage of the innovative development process. This technology may also be applied to other related matters, for example, as supplementary evidence of the existence of a contract or as additional assurance when exchanging information during precontractual negotiations.
WIPO PROOF is an online service that utilizes the user’s local server to create a unique code or “digital fingerprint” for the contents of that file, date- and time-stamped by WIPO. The user receives a token containing said code and WIPO stores a copy of the token on its secure servers in Switzerland. Since files are not uploaded nor stored in external servers, confidentiality of information is not compromised, which is of relevance when dealing with sensitive information. The code can easily be verified online for free, at any time. The system will determine if the code of the original digital file matches the one stored in the token. If the original digital file has been modified, the code will not be verified.
By means of this new service, WIPO offers users a creative and accessible solution to some of the challenges posed by the era of digital transformation.

Personalized medicine and patents
Personalized medicine is based on the administration of active ingredients to individuals sharing a specific biological feature. It relates to targeted therapies that search for the most appropriate treatment for a given patient, and that are usually presented as the promising future of medicine.
In many cases, the active ingredients used in these treatments are known compounds. However, determining the effective doses and the population for which such administration is therapeutically effective may involve experimental developments, as well as considerable economic efforts. In this context, it is of interest to know whether this “personalized medical use” resulting from research can be protected by a patent.
In other words, is it possible to claim a compound X for the treatment of disease Y administered to patients that share feature Z or, alternatively, a treatment for disease Y comprising administering a compound X to patients that share feature Z?
Under the European patent system, the possibility of second medical uses claims is provided by Art 54(5) EPC. A medical use of a known compound X may confer novelty to a claim directed to compound X for the treatment of disease Y, as long as said treatment does not form part of the state of the art. If a related technical effect is demonstrated, the therapeutic indication may be considered as a functional feature of the claim, conferring novelty to the claim in accordance with decision G 2/88 of the Enlarged Board of Appeal.
Later on, in decision G 2/08 of 2010, the Enlarged Board stated that a new mode of administration or dosage regime of compound X, or even the treatment of the same disease Y in a new group of patients, could contribute to the novelty of a medical use even if the general use of compound X for the treatment of disease Y were already known. This decision also set forth the prohibition of “Swiss type” claims for second medical uses under the European system.
In 2019, decision T 694/16 of a Technical Board of Appeal reaffirmed these principles and extended them specifically to personalized medicine. The case at issue was related to a known compound for the treatment of dementia, where the use of such compound in patients presenting a specific biological marker detectable in cerebrospinal fluid was claimed. According to patent EP2170104, this marker allows to distinguish between patients in prodromal phase, who will develop dementia, and patients presenting cognitive symptoms but who will not necessarily develop dementia. For the Board of Appeal, the selection of a subgroup of patients based on a feature Z (the common biological marker) may contribute to the novelty of a personalized medicine claim: the functional relationship between markers that characterize patients and the therapeutic effect which is sought is an essential technical feature of the claim. Moreover, prior art describing the use of compound X for the treatment of a disease Y in a group of patients including the subgroup having feature Z does not inherently or inevitably disclose this functional relationship and does not affect novelty of the claim.
With this decision, the European Patent Office confirms that it is possible to obtain European patents related to personalized medicine, even if the general use of an active principle in a determined treatment is known in the state of the art. To obtain a valid patent, the personalized treatment must also meet the inventive step requirement, meaning that it must not be obviously derivable from the prior art.
In Argentina, according to a Joint Ministry Resolution issued by the Patent Office and the Ministry of Health in 2012, second medical uses are not deemed patentable: nowadays, personalized medicine may hardly be the object of a patent of invention, regardless of whether novelty and inventive step criteria are met. Other Latin American countries accept second medical uses, as long as they are claimed according to local practice and all other patentability requirements are met.
In general, claims directed to uses are not accepted in the United States, although therapeutic methods for treatment are permitted. Nonetheless, the doctrine of inherency and anticipation practice under the US patent system may make it difficult to protect personalized medicine as compared to the European system.

EPO’s (final?) decision on patentability of plants and animals
The Enlarged Board of Appeal of the European Patent Office (EPO) confirmed in its most recent decision that plants and animals exclusively obtained by essentially biological processes are excluded from patentability.
This would be the final chapter in a debate that had the Technical Boards of Appeal of the EPO against the European Commission and the Administrative Council of the European Patent Organisation, and which may be summarised as follows:
- Article 53(b) EPC excludes from patentability essentially biological processes for the production of plants or animals.
- In 2015, the Enlarged Board of Appeal had concluded that this exclusion did not extend to plants or animals exclusively obtained by means of an essentially biological process, according to decisions G 2/12 (“Tomato II”) and G 2/13 (“Broccoli II”).
- In 2017, the European Commission promoted the amendment of Rule 28 EPC by the Administrative Council, based on the interpretation of Directive 98/44/CE, thus explicitly excluding plants and animals obtained exclusively by means of essentially biological processes.
- Recently, a Technical Board of Appeal refrained from applying amended R28, arguing that it would be contrary to the European Patent Convention, referring to the Enlarged Board’s decision of 2015.
- In the face of this legal uncertainty, the President of the EPO referred the question to the Enlarged Board in 2019.
- In Decision G 3/19 “Pepper”, the Enlarged Board changed its position and ruled that the exclusion from Art 53(b) also applies to plants and animals exclusively obtained by means of an essentially biological process.
In reaching this conclusion, the Enlarged Board adopted a dynamic interpretation of the Convention, recognising that the 2017 Rule amendment leads to an interpretation that is contrary to the 2015 decisions.
Furthermore, the Enlarged Board claims that this decision shall not have retroactive effect on European patents granted, or pending applications filed, before 1 July 2017, date on which the amended Rule came into force.
The foregoing would not modify patentability before the EPO of plants and animals that are not exclusively obtained from essentially biological processes. For instance, it would be possible to obtain a European patent for genetically modified plants or animals, as long as they are not obtained from traditional reproduction and selection methods.
